Флуоресцентная ультрамикроскопия
Использование флуоресцентных меток различной природы позволяет существенно увеличить контраст исследуемых структур биологических тканей. Наиболее распространенным методом исследования биотканей с использованием флуоресцентных агентов является конфокальная лазерная сканирующая микроскопия (КЛСМ). Этот метод используется для получения трехмерных изображений биотканей с субклеточным разрешением на глубину до 200–300 мкм и с одномоментной областью визуализации от 0,1 до 0,5 мм.
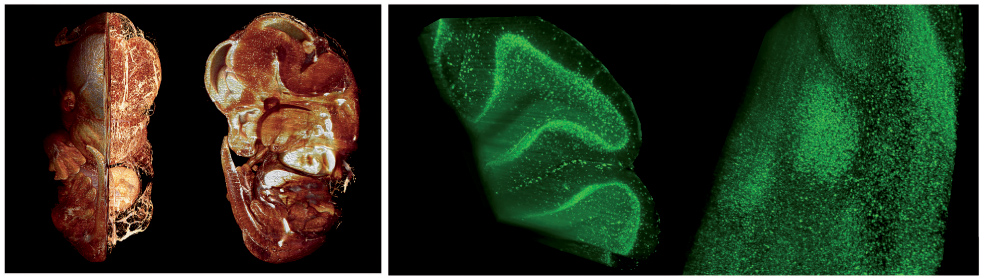
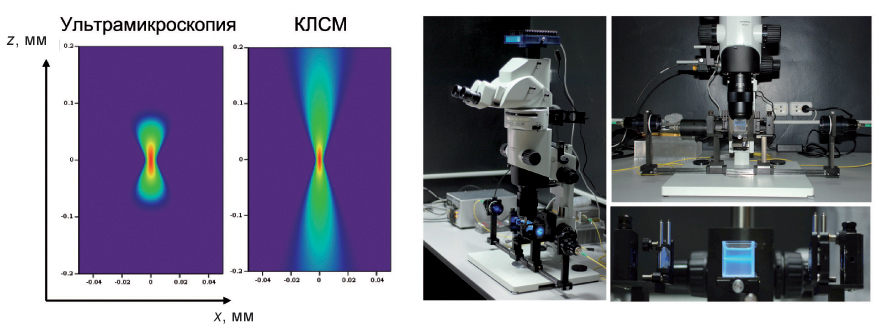
Оптические методы, позволяющие производить визуализацию клеточной структуры предварительно просветленных биологических объектов, органов и тканей в масштабах, сопоставимых с размерами самих этих объектов и органов (1–20 мм), стали появляться только в последние годы. Наиболее перспективным является метод флуоресцентной ультрамикроскопии, принципиальным отличием которого от КЛСМ является то, что флуоресценция возбуждается лишь в узком слое биоткани благодаря использованию специальных систем подсветки, а регистрируется в направлении, перпендикулярном плоскости подсветки. Это позволяет избежать попадания прямого лазерного излучения в приемный тракт системы и паразитного фона засветки вне области исследования: функция размытия точки в ультрамикроскопии гораздо менее вытянута по глубине.
Даже при использовании объективов с небольшой числовой апертурой и большим рабочим расстоянием в таких методах достигается высокое разрешение на больших глубинах в оптически прозрачных образцах. В ИПФ РАН (И. В. Турчин, В. А. Каменский, А. Н. Морозов) совместно с Институтом нормальной физиологии им. П. К. Анохина РАМН разработана установка для флуоресцентной ультрамикроскопии с компактной системой транспортировки лазерного излучения от источника к объекту исследования на основе одномодового волоконнооптического тракта. Для обеспечения высокого разрешения системы формируется тонкий лазерный пучок подсветки на длине волны 488 нм с шириной, соответствующей поперечному размеру исследуемого объекта.
Основные достоинства такого решения по сравнению с открытыми системами – высокое качество пучка подсветки в области исследования из-за близости пространственной моды излучения к гауссовой, невосприимчивость системы к внешним воздействиям (температура, вибрации, пыль), простота использования и перемещения. На созданной установке получены трехмерные изображения предварительно просветленных образцов биологических тканей (легких, сердца, перинатального мозга мышей и плодов мыши) с использованием иммуногистохимической окраски и автофлуоресценции.
Продемонстрировано высокое разрешение метода (5–13 мкм) при больших размерах исследуемых объектов (до 10 мм). Эти параметры существенно превосходят достигаемые в традиционной конфокальной микроскопии. Полученные результаты (В. А. Каменский) имеют большое значение для детального изучения структурных и функциональных особенностей строения органов и тканей в биологии развития, нейробиологии и экспериментальной медицине.