Диффузионная флуоресцентная томография
Флуоресцентный in vivo имиджинг лабораторных животных в последние годы стал широко использоваться в доклинических экспериментальных исследованиях при решении самых разных задач, таких как разработка новых методов диагностики различных патологических процессов, оценка эффективности и выявление механизмов действия терапевтических препаратов, быстрый скрининг новых лекарственных агентов. В экспериментальной онкологии животным имплантируются опухоли, меченные флуоресцентными агентами, что позволяет исследовать развитие первичных опухолей и метастазов на молекулярном уровне in vivo. Для определения размеров такой опухоли, как правило, используется метод поверхностного флуоресцентного имиджинга, при котором исследуемый объект облучается широким световым пучком, возбуждающим флуоресценцию, а слабое флуоресцентное свечение регистрируется на поверхности животного высокочувствительной цифровой камерой. Данный метод позволяет оперативно (за 1–5 с) оценить поперечные размеры новообразования. Однако для глубоко расположенных опухолей точность такой оценки мала, так как свет, проходя сквозь биоткани, сильно рассеивается, и изображение получается сильно размытым.
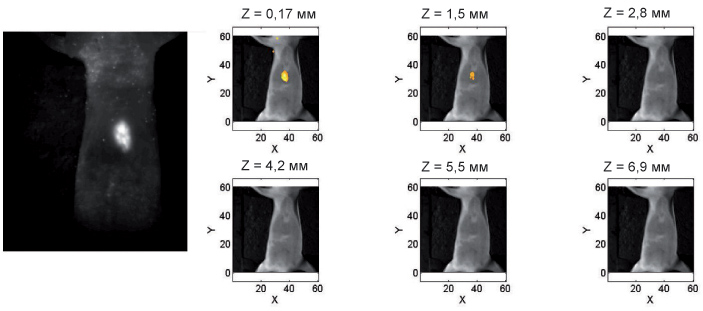
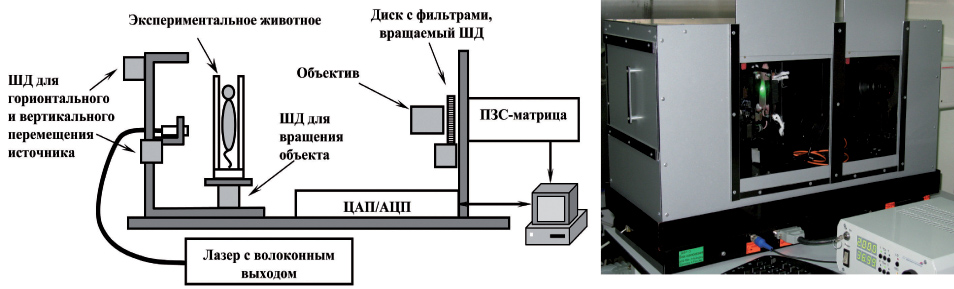
Метод диффузионной флуоресцентной томографии (ДФТ) позволяет проводить трехмерную реконструкцию флуоресцирующих включений, используя информацию о распределении интенсивности флуоресценции на поверхности объекта при различных положениях источника излучения. Поскольку в рассеивающем объекте траектории фотонов являются случайными, а не прямолинейными, как, например, в рентгеновской томографии, задача восстановления распределения флуорофора требует разработки специализированных алгоритмов, учитывающих особенности распространения света в мутных средах. В ИПФ разработана ДФТ-установка с плоской просветной геометрией сканирования исследуемого объекта (И. В. Турчин, И. И. Фикс, М. С. Клешнин, В. А. Каменский). Сканирование ведется механическим перемещением по поверхности животного торца оптоволокна, совмещенного с лазерным источником излучения, возбуждающим флуорофор; флуоресценция регистрируется при каждом положении источника высокочувствительной цифровой камерой с фильтрами, блокирующими возбуждающее излучение.
Для реконструкции трехмерного распределения флуорофора разработан итерационный алгоритм, основанный на функционале Тихонова, позволяющий находить решение системы линейных уравнений при условии неотрицательности его компонент с большей точностью по сравнению с решениями, полученными с помощью алгоритмов общего класса. Для решения прямой задачи в данных алгоритмах применяются разработанные сотрудниками ИПФ гибридные модели светорассеяния в тканях, позволяющие с высокой точностью рассчитывать поле подсветки в рассеивающей среде на произвольном удалении от источника с помощью простых аналитических выражений. Для точного решения уравнения переноса излучения создан программный комплекс на основе метода Монте-Карло, использующий в качестве вычислителя графический процессор. Результаты модельных экспериментов на биоподобных средах показали возможность определить положение центра флуоресцирующего включения и его поперечные размеры с точностью не хуже 1 мм и размеры по глубине с точностью не хуже 1,5 мм. В экспериментах in vivo на лабораторных животных также была продемонстрирована высокая точность метода.
Созданные в ИПФ экспериментальные установки для флуоресцентного имиджинга лабораторных животных были апробированы в совместных биологических экспериментах с ИНБИ РАН, ИБХ РАН, НижГМА, ННГУ и РОНЦ с использованием различных флуоресцирующих агентов – цветных белков, квантовых точек, фотосенсибилизаторов.